The Molekule Air Pro has received clearance from the FDA as a Class II medical device, a classification that was previously approved for the Molekule Air Mini, Molekule Air Mini+ and Molekule Air Pro RX (Read about the FDA clearance process for the Air Mini in this post).
Air Pro was put through the same rigorous FDA process as the Air Mini and Air Pro RX, and the new Class II medical device designation means that Air Pro is now legitimately recognized for its ability to destroy viruses, bacteria and airborne mold for medical purposes.
How the stage was set for Air Pro testing
Extensive research is required for a device to be cleared as a Class II medical device, and Air Pro was no exception. It had to be shown with reasonable assurance that Air Pro is safe and effective in its indications for use, which are capture and destruction of viruses, mold and bacteria. The standard for this is a log 4 reduction or removal of at least 99.99% of target bioaerosols. We worked with the experts at Aerosol Research Engineering Laboratories (ARE) to design a series of experiments that could demonstrate Air Pro was up to the task.
When bacteria, mold, viruses or any other biological particles are floating through the air they are called a bioaerosol. The bioaerosols that are most commonly used for air purifier testing have been extensively applied to tests on Molekule air purifiers in the past, including trials for the Air Pro conducted by Intertek, a multinational assurance, inspection, product testing and certification company. Read more about the Air Pro’s Intertek test results. Because this set of experiments was more geared for Air Mini, the Intertek test data showed the Air Pro’s ability to clean the air actually outperformed the resolution of the air sampling method — but the results were informative, nevertheless.
As a result, the team at ARE spent a lot of time characterizing the air flow and performance characteristics of Air Pro so the experiments could generate data that more accurately showed the model’s effectiveness. They came up with sampling methods that could reliably enumerate the concentration of the different bioaerosols at all time points.
How testing is done with mold, bacteria and viruses
These organisms have characteristics that make them both efficient to work with in a laboratory setting and representative of real-world pollutants. None of them are harmful enough to warrant anything beyond standard safety procedures, and all are very common in nature and at least as hardy as the disease-causing versions from the same kingdom.
MS2 bacteriophage. This virus only infects bacterial cells and is unable to infect human cells, so it is often used as a surrogate for airborne human virus. Given that it attacks bacteria, its individual virus particles are only about .03 microns in size, so they are among the smaller viruses available — even smaller than coronavirus or influenza.

The physical structure of MS2 bacteriophage.
Aspergillus brasiliensis. Though our previous research mentions Aspergillus niger as the testing mold spore, in 2020 it was discovered the Aspergillus niger spore stock used by laboratories around the world was in fact a different species, Aspergillus brasiliensis. Regardless, there are hundreds of Aspergillus species and almost all are black molds that thrive in moist or wet environments. Many mildew colonies are formed from this mold, and it can also be found between the layers of older onions and other vegetables. Aspergillus can cause the occasional allergic reaction or trigger asthma, but it very rarely infects people with severely compromised immune systems. Standard laboratory sterilization procedures are all that is required to keep everyone safe in the testing facilities.

Zoomed in A. brasiliensis, each spore-bearing vesicle is around 50 microns wide. The attached spores are anywhere from 2 to 10 microns.
Escherichia coli. Most of us are familiar with E. coli as gut bacteria that can cause sewage-borne infections. Only a few strains of E. coli are actually infectious — most are benign. E. coli makes a great model for aerosolized bacteria because it can survive in the air for several hours.

E. coli through a microscope. Each cell is about 2 microns long.
Bacillus subtilis. This organism is also very benign and is not known to cause any diseases in humans. What makes this organism useful is that it produces extremely tough endospores that can last for years, even in the vacuum of space. The experiments aerosolized a dry powder composed only of these endospores.

Bacillus subtilis (red rods) and its endospores (green dots). The endospores are about 1 micron wide and 2 microns long.
Measuring with precision
One of the main goals of these experiments was to measure the concentration of hundreds of thousands of bioaerosol particles in the air. The team used sampling and plating techniques with enough resolution to detect a single virus.
First, the natural settling of all the bioaerosols was determined so it could be subtracted from the measured removal of the air purifier. The graphs below are Net Log reductions, which is the reduction after natural settling has been taken into account.
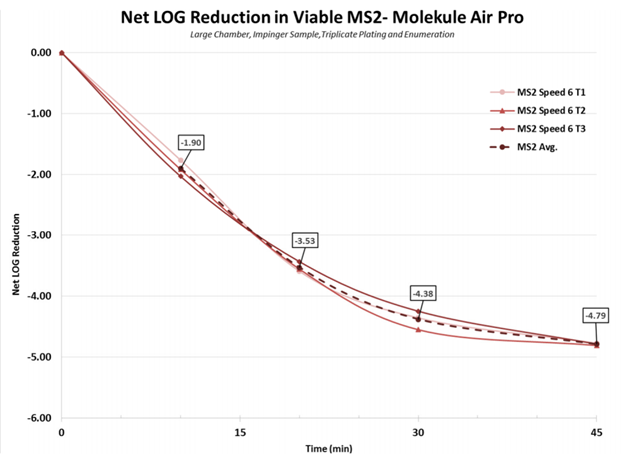
Air Pro can also remove the MS2 bacteriophage quickly, getting past log 4 reduction at 30 minutes and to 4.79 log reduction, or 99.998% removal at 45 minutes.
All of the tests above were done with the Air Pro running at its highest speed of 6. In addition to maximum performance, we were also interested in seeing how the Air Pro performed at lower speeds. To that end, the researchers tested capture of MS2 bacteriophage at speeds 2, 3 and 6, and then compared the reduction rates.

The tests showed that when running at the slower speeds, Air Pro still removes large amounts of virus particles from the air. At speed three, it takes about an hour to get past log 4 and about two hours at speed 2. Even so, at the very quiet speed 2, log 1.31 or 95% of the virus particles were removed within 30 minutes and log 2.61 or 99.75% within the hour.
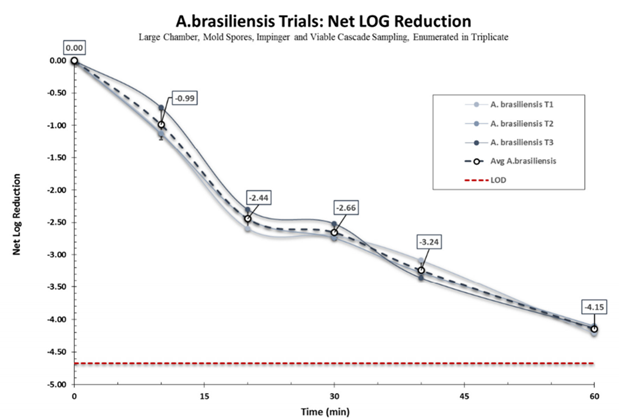
The test to remove Aspergillus spores came in at 4.15 log reduction, or 99.993% removed in one hour after averaging over three trials.

Though the concentration dropped more quickly at the beginning for the E. coli tests, it ended up at roughly the same point on average, 4.2 log reduction or 99.994%.

The bacillus endospores were a little easier for Air Pro to remove, and it achieved log 4 reduction at around half an hour. After the full hour there was a 5.8 log reduction or 99.9998% of the endospores gone.
You can read Air Pro’s whole lab report on the different bioaerosols here, and the whole report on Air Pro removing viruses at different speeds here.
How Air Pro more thoroughly protects air quality
The data above shows that Air Pro can remove bioaerosols from the air very quickly and how Air Pro can achieve the log 4 reduction that we submitted to the FDA as part of our 510(k) Class II medical device clearance application. Quite simply, viruses, mold, bacteria and any of their spores can be captured and destroyed quickly and efficiently by Air Pro.
Air Pro is rated to clean the air in an area up to 1,000 square feet in diameter, four times as much space as Air Mini. It does so by using PECO technology, which are high efficiency filters that not only destroy bioaerosols, but also destroy and capture airborne particles. It also has a sensor suite to detect three sizes of particulate matter in the air. When Auto Protect mode is active, Air Pro will automatically ramp fan speed up and down and adjust to ambient air quality conditions based on the level of particles it detects. Thus, Auto Protect offers a balance of sound and performance, making it ideal for use in homes and workspaces.
At Molekule we are pursuing the science of clean air and communicating about pollution and what to do about it on Molekule’s blog. Subscribe or follow our Facebook, Instagram and Twitter accounts for more information.
A. niger, E. coli, B. subtilus, and MS2 Bacteriophage courtesy of Wikimedia Commons, part of the Wikimedia Foundation.












